Equation 1. Observed rate constant versus pH
Comments
Investigate the effect of changing k0, kH+, kOH-, ka, kb and Ca values on the semi-log plot of observed k' versus pH. Explore the equation by changing the parameter values.
The observed first order rate constant k' may be calculated a function of the specific reaction rate, k0, the specific acid-base catalytic coefficients, kH+ and kOH-, and the general acid-base catalytic coefficients, ka and kb, according to Equation 1.
Equation 1. Observed rate constant versus pH
k0, the specific reaction rate is independent of pH
With specific acid catalysis of the reaction kH+ is multiplied by the concentration of hydrogen ion, H+.
Specific base catalysis is included by multiplying kOH- by the concentration of hydroxyl ion concentration, OH-.
General acid catalysis is included in equation 1 by multiplying ka by the concentration of general acid, HA, given by equation 2.
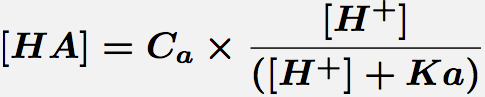
Equation 2. Concentration of the acid form of a monoprotic acid
General base catalysis is included in equation 1 by multiplying kb by the concentration of general base, A-, given by equation 3.
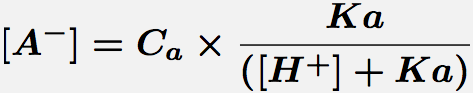
Equation 3. Concentration of the base form of a monoprotic acid