
Figure 19.2.1 Two Compartment Pharmacokinetic Model
return to the Course index
previous | next

Figure 19.2.1 Two Compartment Pharmacokinetic Model
Clearance Model
Equilibrium Model
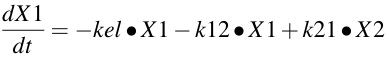
Equation 19.2.1 Differential Equation for the Central Compartment
The kel • X1 term describes elimination of the drug from the central compartment, while the k12 • X1 and k21 • X2 terms describe the distribution of drug between the central and peripheral compartments. Writing differential equations can be reviewed in Chapter 2.
Equation 19.2.2 Integrated Equation for Plasma Concentration versus Time
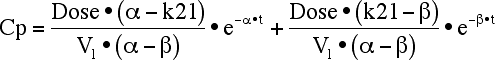
Equation 19.2.3 Integrated Equation for Cp versus Time including k21 and V1
with α > β and
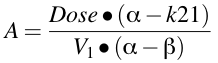
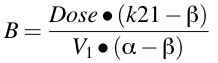
Equation 19.2.4 Calculating values for A and B
The A, B, α, and β terms were derived from the micro-constants during the integration process. They are functions of the micro-constant k12, k21, kel and V1
Using the substitutions for the sum and product of α and β.
α + β = kel + k12 + k21
α • β = kel • k21
If we know the values of kel, k12 and k21 we can calculate α + β as well as α • β. Substituting these values into Equation 19.2.3 gives us values for α and β.
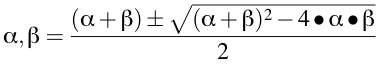
Equation 19.2.5 Converting from kel, k12 & k21 to α & β
Note, in this equation, α is calculated when '+' is used in the numerator and β is calculated when '-' is used in place of the '±'. Thus α is greater than β.
Once we have values for α and β we can calculate values for A and B using Equation 19.2.4.
Since α + β = kel + k12 + k21 = 0.192 + 1.86 + 1.68 = 3.732
and
α x β = kel x k21 = 0.192 x 1.68 = 0.32256
Now:
α = [(a+b) + sqrt((a+b)2 - 4xaxb)]/2 = [3.732 + sqrt(3.7322 - 4x0.32256)]/2 = [3.732 + 3.5549]/2 = 3.643 hr-1
β = [3.732 - 3.5549]/2 = 0.08853 hr-1
A = Dose x (α - k21)/[V1 x (α - b)] = 500 x (3.643 - 1.68)/[90.5 x 0.39 x (3.643 - 0.08853)] = 500 x 1.963/[35.295 x 3.55447] = 7.824 mg/L
B = Dose x (k21 - b)/[V1 x (α - b)] = 500 x (1.68 - 0.08853)/[35.295 x 3.55447] = 6.343 mg/L
The last step is
Cp = α x e(-α x t) + β x e(-β x t) = 7.824 x e(-3.643 x 1.5) + 6.343 x e(-0.08853 x 1.5) = 7.824 x 0.004234 + 6.343 x 0.8756 = 0.0331 + 5.5542 = 5.59 mg/L
Later in this chapter we will use equations for the reverse process of converting α, β, A and B into values for k12, k21, kel and V1.
Material on this website should be used for Educational or Self-Study Purposes Only
Copyright © 2001 - 2026 David W. A. Bourne (david@boomer.org)
 |
The Book Pharmacokinetics This Course in ePub format |
 |