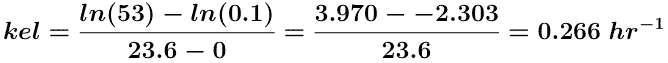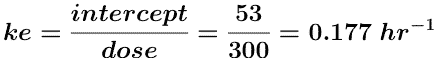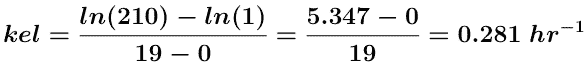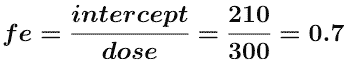Chapter 5
Analysis of Urine Data
return to the Course index
previous | next
Example Calculation of Urine Analysis Plots
After an IV dose of 300 mg, total urine samples were collected and assayed
for drug concentration. Thus the data collected is the volume of urine collected and the drug concentration in urine during each interval. These are the data in columns 1, 2 and 3 of Table 5.6.1.
Table 5.6.1. Example Data Analysis of Drug in Urine Data
Time Interval
(hr)
|
Urine Volume
(ml)
|
Urine Concentration
(mg/ml)
|
Amount Excreted
ΔU (mg)
|
Cumulative Amount Excreted
U (mg)
|
Midpoint Time
tmidpt (hr)
|
Rate of Excretion
ΔU/Δt (mg/hr)
|
A.R.E.
(mg)
|
|
0 - 2
|
50
|
1.666
|
83.3
|
83.3
|
|
|
116.8
|
|
|
|
|
|
|
1
|
41.7
|
|
|
2 - 4
|
46
|
1.069
|
49.2
|
132.5
|
|
|
67.7
|
|
|
|
|
|
|
3
|
24.6
|
|
|
4 - 6
|
48
|
0.592
|
28.4
|
160.9
|
|
|
39.2
|
|
|
|
|
|
|
5
|
14.2
|
|
|
6 - 8
|
49
|
0.335
|
16.4
|
177.3
|
|
|
22.8
|
|
|
|
|
|
|
7
|
8.2
|
|
|
8 - 10
|
46
|
0.210
|
9.7
|
187
|
|
|
13.2
|
|
|
|
|
|
|
9
|
4.8
|
|
|
10 - 12
|
48
|
0.116
|
5.6
|
192.5
|
|
|
7.6
|
|
|
|
|
|
|
11
|
2.8
|
|
|
12 - 18
|
134
|
0.047
|
6.3
|
198.8
|
|
|
1.3
|
|
|
|
|
|
|
15
|
1
|
|
|
18 - 24
|
144
|
0.009
|
1.3
|
200.1
|
|
|
-
|
|
|
|
|
|
|
21
|
0.2
|
|
|
24 - ∞
|
-
|
-
|
-
|
200.1
|
|
|
|

|
Completing the Table
- Multiplying column 2 by column 3 gives column 4, the amount excreted during the interval, ΔU.
- Accumulating the ΔU amounts in column 4 gives the cumulative amount excreted, U, up to the end of the current time interval. The total amount excreted, U∞ is found at the bottom of column 5, that is 200.1 mg.
- The rate of excretion, ΔU/Δt, is calculated by dividing the ΔU amount by the length of the time interval, Δt.
- A.R.E. in the last column is calculated by subtracting the value for U in each row from U∞.
The Data Plots
Cumulative Amount Excreted into Urine Plot
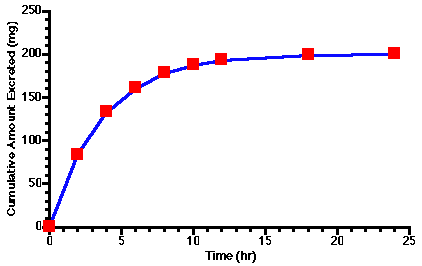
Figure 5.6.1 Linear Plot of Cumulative Amount Excreted versus Time
The plot in Figure 5.6.1 shows U rapidly increasing at first then leveling off to U∞ (= 200 mg). NOTE: U∞ ≠ DOSE for this set of data. Notice that U∞/2 (100 mg) is excreted in about 3 hours which gives an estimate of the elimination half-life. Otherwise this plot is a qualitative representation of the data.
Calculation Using Rate of Excretion Data
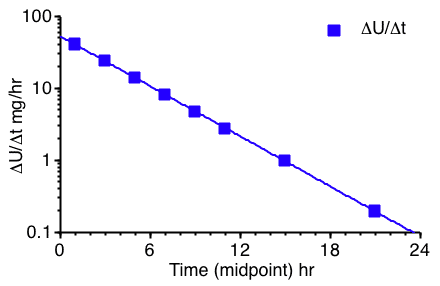
Figure 5.6.2 Semi-log Plot of Rate of Excretion versus Time
The plots in Figures 5.6.2 and 5.6.3 (below) are more useful for calculating parameter values. A straight line can be drawn through the data on each semi-log plot. The elimination rate constant, kel, can be determined from the slope of this line and ke or fe determined from the intercept.
Figure 5.6.2 provides a semi-log plot of ΔU/Δt versus t
midpoint. As you can see this gives a reasonably straight line plot.
Estimating the intercept value to be 53 mg/hr and if the line crosses the axis at 23.6 hr where the rate of excretion is 0.1 mg/hr a value for kel can be estimated.
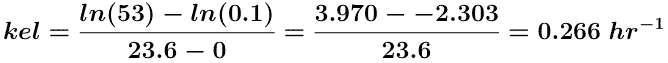
and ke can be determined from the intercept.
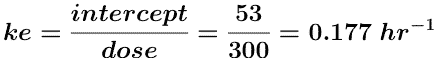
Thus fe = ke/kel = 0.177/0.266 = 0.665.
This plot can be used to estimate kel, ke and fe. A disadvantage of this type of plot is that the error present in "real" data can obscure the straight line and lead to results which lack precision. Also it can be difficult to collect frequent, accurately timed urine samples. This is especially true when the elimination half-life is small.
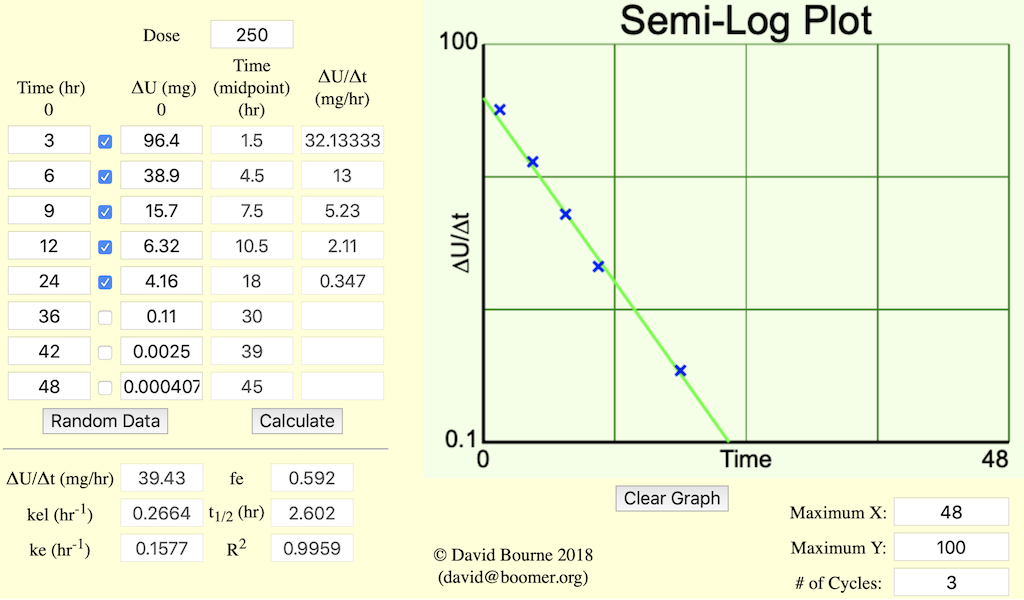
Figure 5.6.3 Analysis of Urine Data - Rate of Excretion
Calculation Using A.R.E. Data
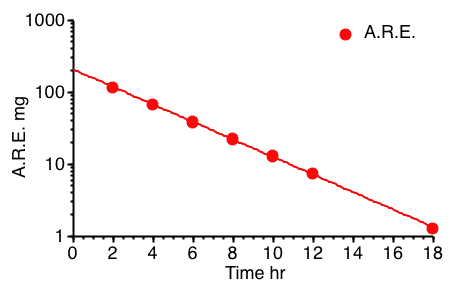
Figure 5.6.4 Semi-log Plot of A.R.E. versus Time
The A.R.E. data are plotted as red circles on Figure 5.6.3 above. Estimating the intercept value to be 210 mg and if the line crosses the axis at 19 hr where the A.R.E. is 1 mg a value for kel can be estimated.
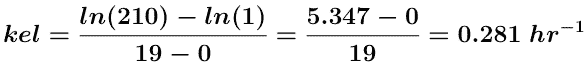
and fe can be determined from the intercept.
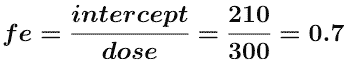
Thus ke = fe • kel = 0.7 x 0.281 = 0.197 hr-1
One disadvantage of this approach is that the errors are
cumulative, with collection interval, and the total error is
incorporated into the U∞ values and therefore into each A.R.E. value. Another problem is that total (all) urine collections are necessary. One
missed sample means errors in all the results calculated.
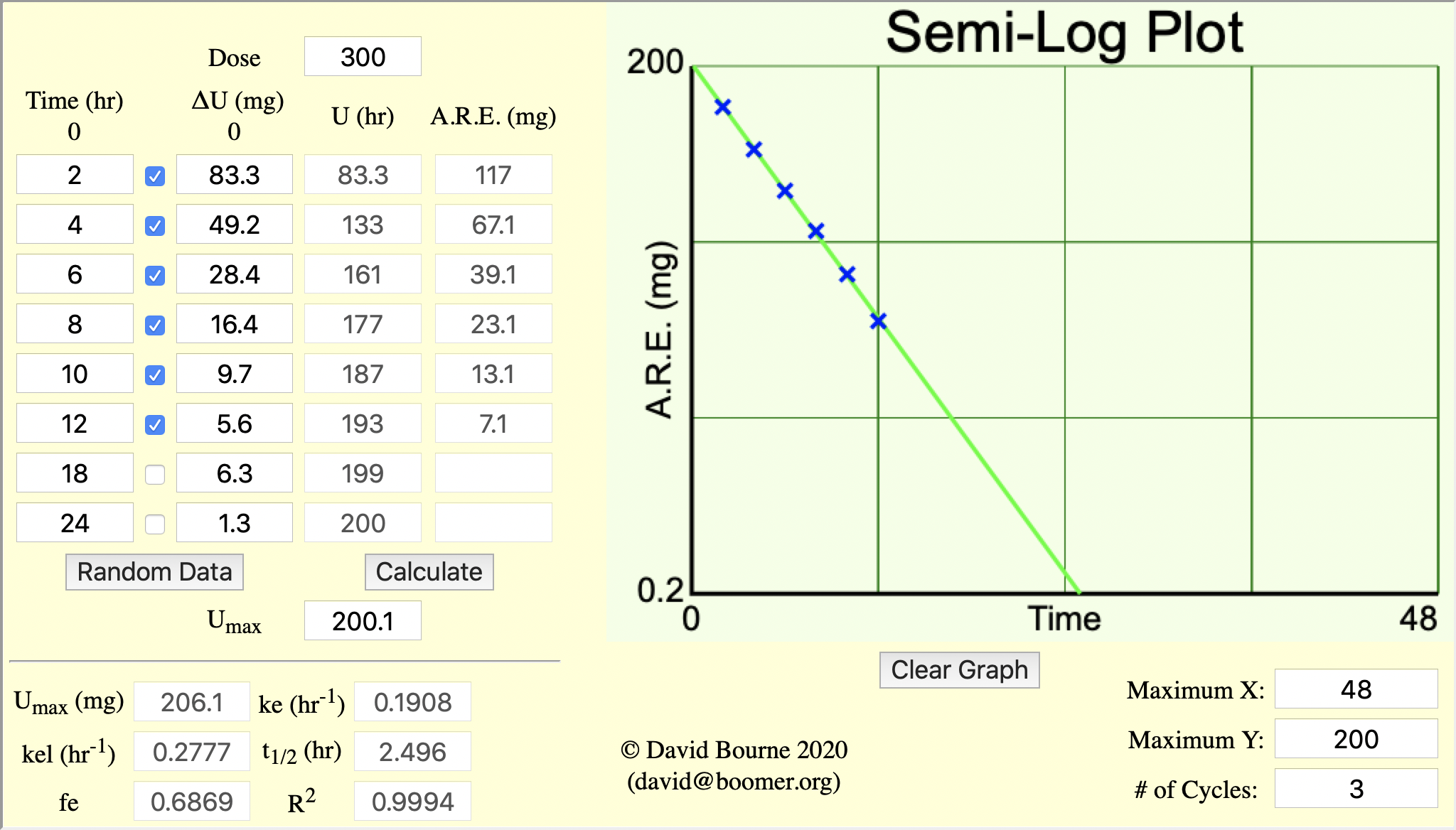
Figure 5.6.5 Analysis of Urine Data - Amount Remaining to Excreted
For practice you can use these urine data collected after an IV bolus dose and estimate pharmacokinetic parameters.

Student Objectives for this Chapter
return to the Course index
This page was last modified: Sunday, 28th Jul 2024 at 4:48 pm
Privacy Statement - 25 May 2018
Material on this website should be used for Educational or Self-Study Purposes Only
Copyright © 2001 - 2026 David W. A. Bourne (david@boomer.org)
 | Survey Help
An iPhone app that allows the input of
up to four locations (latitude and longitude)
and provides the user's distance from each location |

|